Background
|
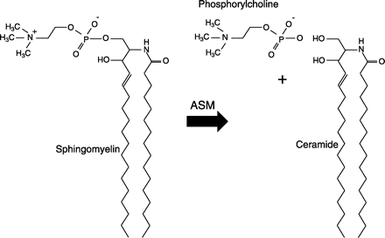
Niemann-Pick disease is caused by mutations in SMPD1, which is important in converting sphingomyelin into ceramide. Patients with Niemann-Pick type B have 10% the normal level of ASM, which is used in this conversion (Figure 1). Ceramide is needed for a variety of processes in the body. Some main roles it plays are in programmed cell death, cell proliferation, cell differentiation, and light sensitivity [1]. There is only about 10% normal levels of ceramide to function in all these processes [2]. I decided to focus on light sensitivity for my future research because not much has been looked at in relation to light sensitivity and Niemann-Pick type B.
|
One symptom of Niemann-Pick diesase that people suffer from is sleep inversion. This means they are awake at night and asleep during the day, which could be caused by a problem with light sensitivity. I found this to be really interesting and wanted to investigate more. I started by looking at proteins that are known to interact with SMPD1 to see if any played a role in sleep. In humans all the interacting proteins were only known to play a role in apoptosis or catabolism. Next I decided to look at interacting proteins in Drosophila as they have a similar phenotype to humans with Niemann-Pick. In Drosophila I found a protein Lazaro that interacts with the fly homolog of SMPD1, CG3376. This protein is known to play a role in light sensitivity and phototransduction. Since sleep inversion definitely seems like it is caused by a problem with light sensitivity, I decided to investigate Lazaro further for my research.
There is no direct homolog of Lazaro in other organisms, so I decided to look at the family that is in Lazaro. Lazaro shares a family with the protein Wunen, which also interacts with CG3376. Wunen does have a direct homolog in mice and humans, which is PPAP2A. This protein is known to play a role in lipid metabolism, but it is unknown what else it functions in [3]. I want to see if it functions in light sensitivity since it shares a family with Lazaro. This led me to my first question:
There is no direct homolog of Lazaro in other organisms, so I decided to look at the family that is in Lazaro. Lazaro shares a family with the protein Wunen, which also interacts with CG3376. Wunen does have a direct homolog in mice and humans, which is PPAP2A. This protein is known to play a role in lipid metabolism, but it is unknown what else it functions in [3]. I want to see if it functions in light sensitivity since it shares a family with Lazaro. This led me to my first question:
Where are SMPD1 and PPAP2A expressed in cells?
My hypothesis for this question is that wherever these proteins are located, if there are low levels of SMPD1, then there will also be low levels of PPAP2A since it is downstream of Lazaro. For my experiment I want to do a microarray to look at expression levels in different tissues in the body [4]. I will look at liver, lung, and retinal tissues for expression of both proteins in healthy mice and in mice mutant for SMPD1. I chose liver and lung tissues because known phentoypes suggest the presence of SMPD1 in these cells. I chose retinal tissues because I believe these proteins play a role in light sensitivity in the eyes, so I am expecting them to be present in these cells. Expression should be higher for both proteins in healthy mice as compared to the mutant mice. If the proteins are present in all these tissues I can then focus on retinal tissues for my second question:
How does light affect SMPD1 and PPAP2A levels?
My hypothesis for this question is that levels of SMPD1 and PPAP2A will be higher in light versus dark environments. For this experiment I want to do a 2D gel-mass spectrometry analysis to look at expression during different events in the cells. I am looking for the presence of both these proteins in retinal tissue cells. The mass spectrometry results will show the intensities of the proteins in the different situations [5]. Using expression of PPAP2A in healthy mice at night as the threshold, I'm expecting PPAP2A expression to be higher in healthy mice exposed to light. I'm also expecting that in the mice mutant for SMPD1 there will be low expression in both light and dark environments, but there will be slightly higher expression in the light than the dark environment. The conclusion that can be drawn from this is that PPAP2A does play a role in light detection and is affected by the low levels of ceramide found in Niemann-Pick patients. This information is important for developing a potential drug therapy treatment to help fix the symptoms of sleep inversion.
Future Research
1) Do zebrafish with mutant SMPD1 experience similar problems in their gills as humans do in their lungs?
2) What is the relationship between NPC1 (the protein that causes Niemann-Pick type C) and SMPD1 that causes patients to have similar phenotypes?
3) Perform a genome-wide RNAi screen in C.elegans for lipid metabolism genes
2) What is the relationship between NPC1 (the protein that causes Niemann-Pick type C) and SMPD1 that causes patients to have similar phenotypes?
3) Perform a genome-wide RNAi screen in C.elegans for lipid metabolism genes
Here is a copy of my final presentation I gave about Niemann-Pick disease:
| Final Presentation File | |
| File Size: | 799 kb |
| File Type: | |
References
[1] Smith, E., & Schuchman, E. (2008). The unexpected role of acid sphingomyelinase in cell death and the pathophysiology of common diseases. The
FASEB Journal, 22(10), 3419. doi: 10.1096/fj.08-108043
[2] http://www.nnpdf.org/npdisease_01.html
[3] http://www.phosphosite.org/proteinAction.do;jsessionid=42CBF10E7131847DE1720F6046DD3C84?id=25705062&showAllSites=true
[4] Brown, P., & Botstein, D. (1999). Exploring the new world of the genome with DNA microarrays. Nature America Inc. Retrieved from http://gen677.weebly.com/uploads/8/6/5/7/865764/brownbotstein1999microarrayreview.pdf
[5] Fey, S., & Larsen, P. (2001). 2D or not 2D. Current Opinion in Chemical Biology, 5(1), 26. Retrieved from http://gen677.weebly.com/uploads/8/6/5/7/865764/fey2dgelreview2001.pdf
STRING
[1] Smith, E., & Schuchman, E. (2008). The unexpected role of acid sphingomyelinase in cell death and the pathophysiology of common diseases. The
FASEB Journal, 22(10), 3419. doi: 10.1096/fj.08-108043
[2] http://www.nnpdf.org/npdisease_01.html
[3] http://www.phosphosite.org/proteinAction.do;jsessionid=42CBF10E7131847DE1720F6046DD3C84?id=25705062&showAllSites=true
[4] Brown, P., & Botstein, D. (1999). Exploring the new world of the genome with DNA microarrays. Nature America Inc. Retrieved from http://gen677.weebly.com/uploads/8/6/5/7/865764/brownbotstein1999microarrayreview.pdf
[5] Fey, S., & Larsen, P. (2001). 2D or not 2D. Current Opinion in Chemical Biology, 5(1), 26. Retrieved from http://gen677.weebly.com/uploads/8/6/5/7/865764/fey2dgelreview2001.pdf
STRING